Sentinel’s routine querying tools allow users to:
- Identify potential medical product safety concerns
- Characterize populations of interest
- Characterize the use of medical products
- Perform epidemiologic analyses to assess their impact on health outcomes
Available Routine Querying Tools
Sentinel’s routine querying tools fall into four categories:
Signal Identification Analyses
Signal identification analyses detect new and unsuspected potential safety concerns.
Level 1 Analyses
Level 1 analyses are descriptive analyses.
Level 2 Analyses
Level 2 analyses are retrospective inferential analyses.
Level 3 Analyses
Level 3 analyses are prospective sequential inferential analyses.
Learn more about Sentinel’s routine querying tools:
Routine Querying System, also known as Query Request Package (QRP), is comprised of cohort identification and analytic modules
Routine Querying System Reporting Tool produces tables and figures using the files created by using QRP
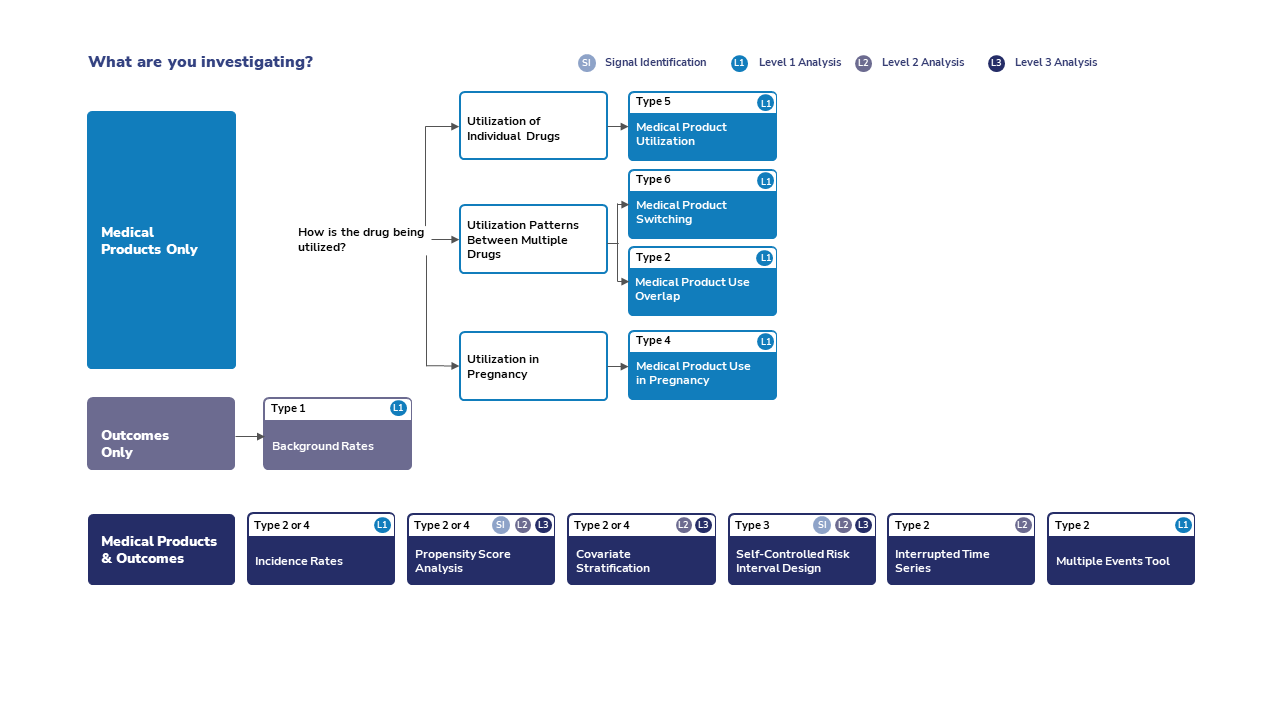
What Routine Querying Tool Should You Use?

The routine querying tool you use depends on what you're investigating.
Routine Querying Tools for Medical Products Only
The tool you use for investigating medical products (only) depends on how the drug is being used.
Utilization of Individual Drugs
If you are investigating medical product utilization, use level 1 analysis, type 5.
Utilization Patterns Between Multiple Drugs
If you are investigating medical product switching, use level 1 analysis, type 6.
If you are investigating medical product use overlap, use level 1 analysis, type 2.
Utilization in Pregnancy
If you are investigating medical product use in pregnancy, use level 1 analysis, type 4.
Routine Querying Tools for Outcomes Only
If you are investigating background rates, use level 1 analysis, type 1.
Routine Querying Tools for Medical Products & Outcomes
If you are implementing a signal identification analysis, you may use a self-controlled risk interval design with a type 3 or you may use a cohort design and compare a treatment group with a referent group using propensity score adjusted analysis. If your cohort is composed of pregnant women, then a type 4 is used. If your cohort is composed of the general population, then a type 2 is used.
If you are implementing a retrospective Level 2 analysis to investigate a specific health outcome, you may use a self-controlled risk interval design with a type 3 or you may use a cohort design and compare a treatment group with a referent group. The comparison can be adjusted via propensity score or covariate stratification. If your cohort is composed of pregnant women, then a type 4 is used. If your cohort is composed of the general population, then a type 2 is used.
If you are implementing a prospective sequential Level 3 analysis, you may use a self-controlled risk interval design with a type 3 or you may use a cohort design and compare a treatment group with a referent group. The comparison can be adjusted via propensity score or covariate stratification. If your cohort is composed of pregnant women, then a type 4 is used. If your cohort is composed of the general population, then a type 2 is used.
If you are implementing an interrupted time series analysis, you may use a level 2 analysis, type 2.
Interested in learning more about routine querying tools? Below is a full list of Sentinel's routine querying tools.
Enhancements to Sentinel's Routine Querying System
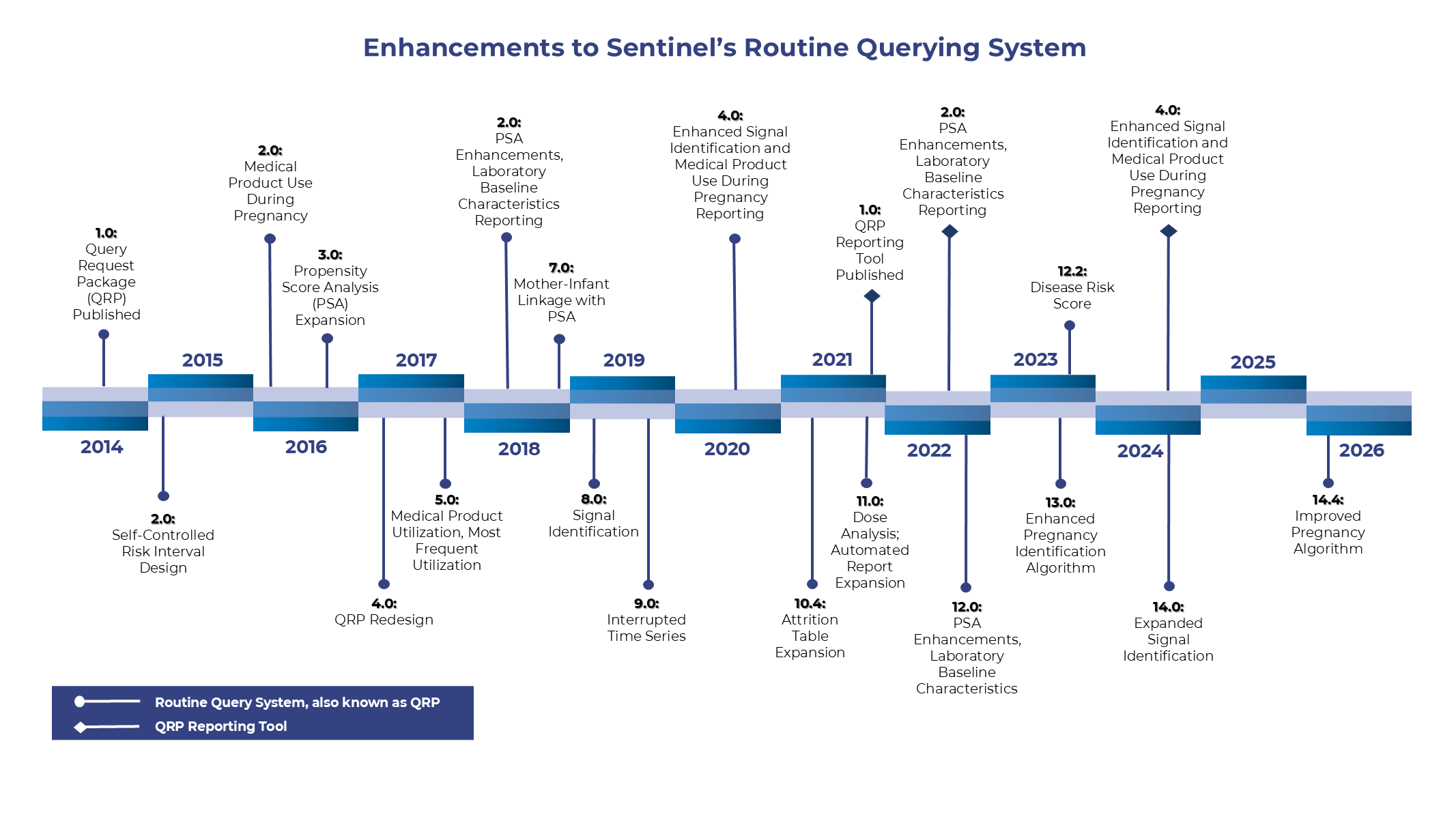
Over time, Sentinel releases enhancements to its routine querying system. These enhancements expand, improve, and refine study design capabilities. For a full description of each enhancement, please see here: Enhancements to Sentinel's Routine Querying System.